Key words
Abbreviations used
Introduction
Case presentation

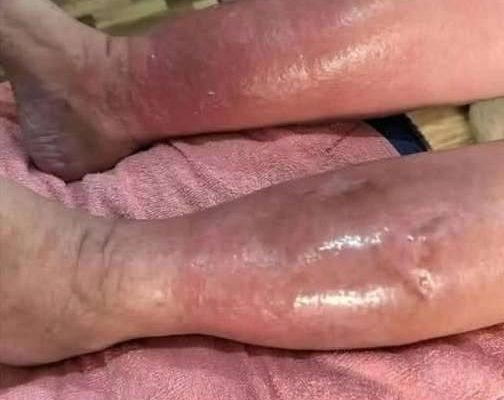
Fig 1. Initial presentation of the patient’s lesions. A, Diffuse erythema and swelling of bilateral hands. B, Red, poikilodermatous plaques with dusky scale in a photodistribution on the neck. C, Tense vesicle on the right ulnar wrist. Histopathology findings of the biopsied lesion are shown in Fig 2.

Fig 2. Histopathology of the vesicle on the right ulnar wrist. A, Intraepidermal vesiculation with necrosis and exocytosis of lymphocytes, histiocytes, neutrophils, and eosinophils. Hematoxylin and eosin, original magnification ×40. B, Interface dermatitis with basal vacuolization and dyskeratosis, lymphohistiocytic infiltrate, and basket-weave orthokeratosis. Hematoxylin and eosin, original magnification ×40.

Fig 3. Progression of vesiculobullous eruption. A, Diffuse erythema with semi-tense vesicles on the dorsal feet and ankles. B, Dusky purple plaques with scale in the bilateral popliteal fossae, with erosions on the right calf from minimal trauma.
Discussion
Table I. Patient demographic characteristics, oncologic history, dermatological clinical and pathologic features, management, and clinical course
| Empty Cell | Case 15 | Case 25 | Our case |
|---|---|---|---|
| Age/gender | 98/F | 68/M | 55/M |
| Diagnosis | MZL → DLBCL | FL → DLBCL | Relapsed FL |
| Previous treatment | R-CHOP, BR, ibrutinib | R-CHOP, R-GemOx, radiation | Rituximab, obinutuzumab, bendamustine, tazemetostat |
| Treatment at time of reaction | LT monotherapy | LT monotherapy | LT and polatuzumab vedotin |
| Time to onset of vesicular rash | 3 cycles (7 weeks) | 4 cycles (12 weeks) | 3 cycles (8 weeks) |
| Other skin manifestations | Preceding photodistributed rash involving other sites; new-onset bilateral lower extremity edema | Preceding photodistributed rash involving other sites; worsening of existing bilateral lower extremity edema | Preceding photodistributed nonpruritic rash on head/neck spreading caudally; new-onset lower extremity edema |
| Biopsy H&E | Subepidermal vesicular dermatitis, no dermal edema | Subepidermal vesicular dermatitis, interface dermatitis, no dermal edema | Vesiculobullous dermatitis, interface dermatitis, no dermal edema |
| Biopsy DIF | Fine granular IgA at basement membrane | Fine grains of IgA alone basement membrane zone | Focal dot-like/granular IgA along basement membrane zone |
| Management | Supportive | Supportive | Topical clobetasol, prednisone 0.5 mg/kg × 1 wk |
| Treatment status/outcome | Discontinued with resolution of symptoms, no recurrence | Discontinued, patient deceased | Discontinued with improvement in symptoms, no recurrence |
Conflicts of interest
References
- 1
A phase I study of ADCT-402 (loncastuximab tesirine), a novel pyrrolobenzodiazepine-based antibody–drug conjugate, in relapsed/refractory B-cell non-Hodgkin lymphomaClin Cancer Res, 25 (2019), pp. 6986-6994, 10.1158/1078-0432.CCR-19-0711
- 2
Loncastuximab tesirine in relapsed or refractory diffuse large B-cell lymphoma (LOTIS-2): a multicentre, open-label, single-arm, phase 2 trialLancet Oncol, 22 (2021), pp. 790-800, 10.1016/S1470-2045(21)00139-X
- 3
The antibody-drug conjugate loncastuximab tesirine for the treatment of diffuse large B-cell lymphomaBlood, 140 (2022), pp. 303-308, 10.1182/blood.2021014663
- 4
The diverse landscape of dermatologic toxicities of non-immune checkpoint inhibitor monoclonal antibody-based cancer therapyJ Cutan Pathol, 50 (2023), pp. 72-95, 10.1111/cup.14327
- 5
Blistering lesions associated with loncastuximab tesirineJAMA Dermatol, 158 (2022), pp. 831-832, 10.1001/jamadermatol.2022.1389
- 6
Tazemetostat for patients with relapsed or refractory follicular lymphoma: an open-label, single-arm, multicentre, phase 2 trialLancet Oncol, 21 (2020), pp. 1433-1442, 10.1016/s1470-2045(20)30441-1
- 7
Phase 1b open-label study of loncastuximab tesirine in combination with other anticancer agents in patients with relapsed/refractory B-cell non-Hodgkin lymphoma (LOTIS-7)J Clin Oncol, 41 (2023), Article TPS7581, 10.1200/JCO.2023.41.16_suppl.TPS7581
- 8
Incidence, onset, and management of edema and effusion in patients treated with loncastuximab tesirine for R/R DLBCL in the LOTIS clinical trial programClin Lymphoma Myeloma Leuk, 21 (2021), pp. S397-S398
- 9
Camidanlumab tesirine in patients with relapsed or refractory lymphoma: a phase 1, open-label, multicentre, dose-escalation, dose-expansion studyLancet Haematol, 8 (2021), pp. e433-e445, 10.1016/s2352-3026(21)00103-4
- 10
Rovalpituzumab tesirine, a DLL3-targeted antibody-drug conjugate, in recurrent small-cell lung cancer: a first-in-human, first-in-class, open-label, phase 1 studyLancet Oncol, 18 (2017), pp. 42-51, 10.1016/s1470-2045(16)30565-4
Cited by (2)
-
Dermatologic adverse events associated with antibody-drug conjugate loncastuximab: A retrospective multicenter cohort study
2026, Journal of the American Academy of Dermatology
-
Funding sources: None.
-
Patient consent: The authors obtained written consent from the patient for their photographs and medical information to be published in print and online, with the understanding that this information may be publicly available. Patient consent forms were not provided to the journal but are retained by the authors.